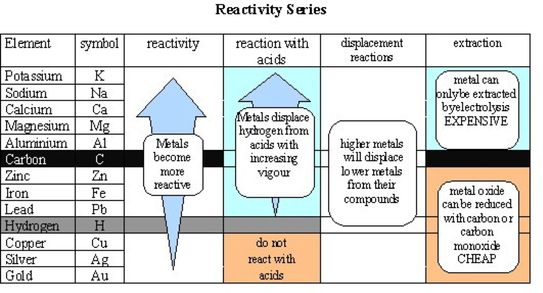
The total energy for the process is the sum of the energies of the individual steps.Write the net balanced equation for this reaction that includes all of the steps above.second ionization energy (ga s) (+1733):.first ionization energy (gas) (+906 kJ/mol):.For zinc, write the chemical equations for the individual steps:.There are several steps to get from metal in the solid state to Zn 2+(aq). We predict that placing a strip of zinc metal in a copper (II) sulfate solution will produce metallic copper and zinc sulfate. For example, zinc is above copper in the series. We can use the series to predict whether a metal displacement reaction will occur. So this is really more than the loss of electrons. The activity series allows us to predict whether a metal displacement reaction will occur. Zinc is defined as reacting vigorously with H+ in aqueous solution. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
